
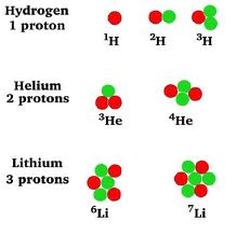
Radium was first discovered in 1898 by Marie and Pierre Curie, and it served as the basis for identifying the activity of various radionuclides.
One curie of activity equals the rate of radioactive decay of one gram (g) of radium-226.
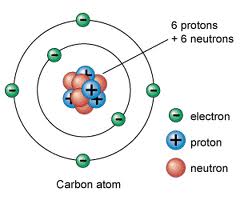
Radium is a radioactive element that occurs naturally in very low concentrations
(about one part per trillion) in the earth’s crust. Radium in its pure form is a silvery-white
heavy metal that oxidizes immediately upon exposure to air. Radium has a density about onehalf
that of lead and exists in nature mainly as radium-226, although several additional isotopes are present.
Click Here to Read the Instructions for the Experiment on Monday 26/11.
One curie of activity equals the rate of radioactive decay of one gram (g) of radium-226.
Radium is a radioactive element that occurs naturally in very low concentrations
(about one part per trillion) in the earth’s crust. Radium in its pure form is a silvery-white
heavy metal that oxidizes immediately upon exposure to air. Radium has a density about onehalf
that of lead and exists in nature mainly as radium-226, although several additional isotopes are present.
Click Here to Read the Instructions for the Experiment on Monday 26/11.



 RSS Feed
RSS Feed
